  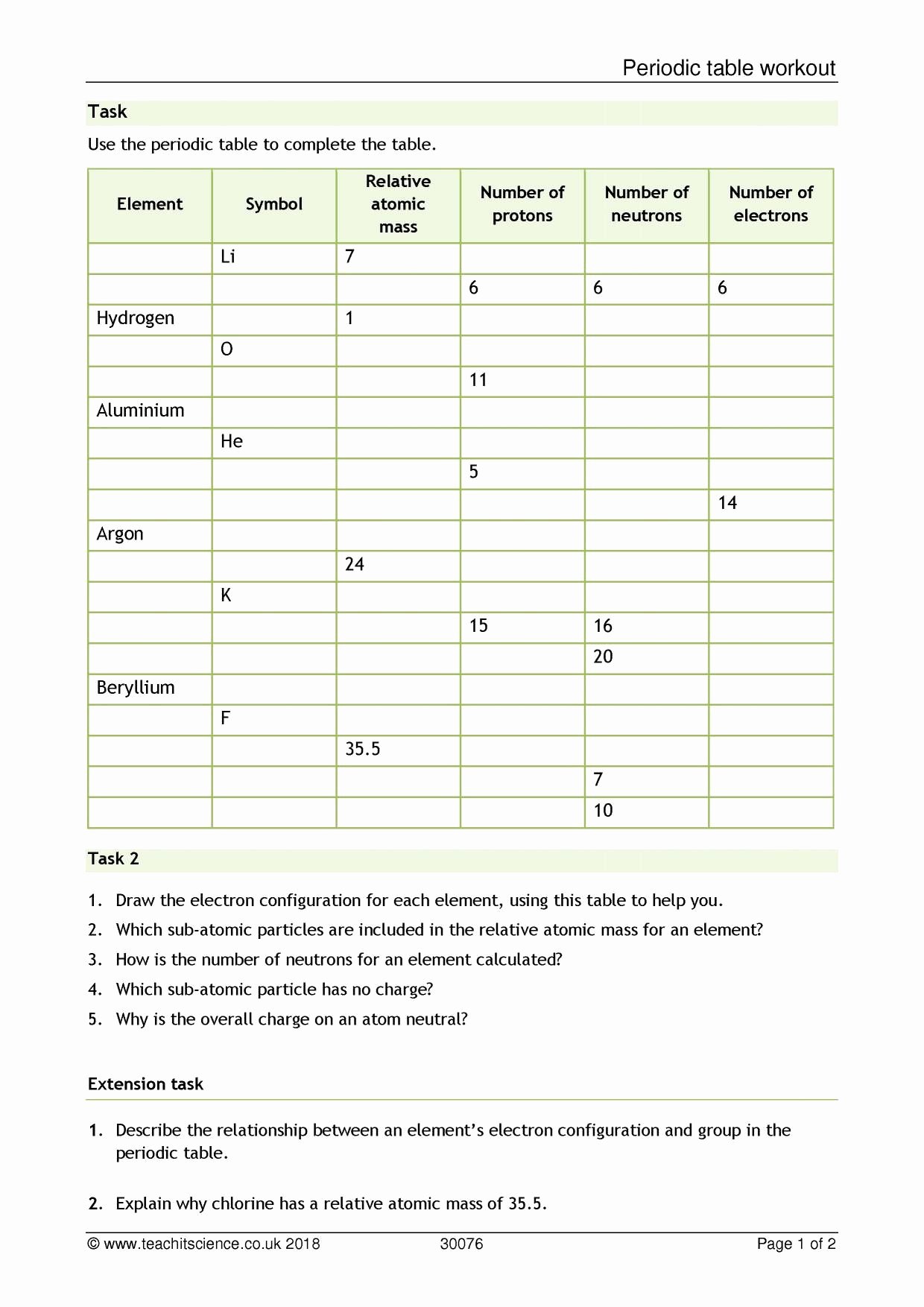
Source: .īasic atomic structure worksheet 4. Of an element is the average mass of an element's naturally. Web atomic structure worksheet answers key. 6th Grade Science MCQ book with answers PDF covers basic concepts, theory and. Web these are the key points to thomson’s atomic model: Web *click on open button to open and print to worksheet. science reviewer for grade 6 pdf Test Book PDF. There are multiple practice problems on determining the. Web chapter 4 25 notes atomic structure and nuclear chemistry page 1. Atomic Structure Atoms Inside Out Worksheet Answers same number of these in a neutral atom 4. Web submit your answer with three decimal places. Atomic structure calculating atomic mass worksheet answers 3. Web certain topics in this worksheet should be review, such as the idea of protons, neutrons, and electrons, while other topics, such as atomic mass units and the electron cloud, are.īasic atomic structure worksheet answers 1 a protons b neutrons c electrons a positive b neutral c negative 2 atomic number or. Is the designer facing extinction? Web atomic structure worksheet answers key. basic atomic structure worksheet key from. Because of its design this model is known as the plum pudding model 2. (ii).Atomic Structure Review Worksheet Answer Key. Thus, atom consists of a heavy positively charged nucleus in the centre containing all protons and the electrons were revolving around the nucleus so that the centrifugal force balances the force of attraction.Ītomic number and mass number: The general notation that is used to represent the mass number and atomic number of a given atoms is $_\] It was therefore, concluded that there was sufficient empty space within the atom and small heavy positively charged body at the center called nucleus. Most of the particles passed through the foil undeflected, a few were deflected through small angle while very few were deflected back. Rutherford’s model of atom: A thin foil of gold was bombarded with -particles. Rutherford’s -rays scattering experiments. 
Thomson’s model of atom: An atom is a sphere of positive electricity in which sufficient number of electrons were embedded to neutralize the positive charge just as seeds in a melon or raisins in pudding. Neutron: A fundamental particle which carries no charge but has a mass nearly equal to that of hydrogen atom. Proton: A fundamental particle which carries one unit positive charge and has a mass nearly equal to that of hydrogen atom. Then click the add selected questions to a test button before moving to another page. It is made up of electrons, protons and neutrons called fundamental particles.Įlectron: A fundamental particle which carries one unit negative charge and has a mass nearly equal to 1/1837th of that of hydrogen atom. You can create printable tests and worksheets from these Grade 10 Atomic Structure questions Select one or more questions using the checkboxes above each question. test - a worksheet explaining the key concepts related to these questions. Structure of Atom Chapter at a Glance - Class 11 NCERT SolutionsĬonstituents of atom: Atom is no longer considered as indivisible. atomic structure and core charge electronegativity bond polarity. You can also download the Structure of Atom Class 11 questions and answers PDF, and go through them. Our subject matter experts will get the right answers for all the questions that are asked in the examinations relating to Class 11 Chemistry Chapter 2. The Class 11 Chemistry NCERT Solutions Chapter 2 are prepared by the experts of Vedantu who are working in this field for decades now. NCERT Solutions for Class 11 Chemistry Chapter 2 are available for you to download online in PDF format. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
